Glucosamine Sulfate
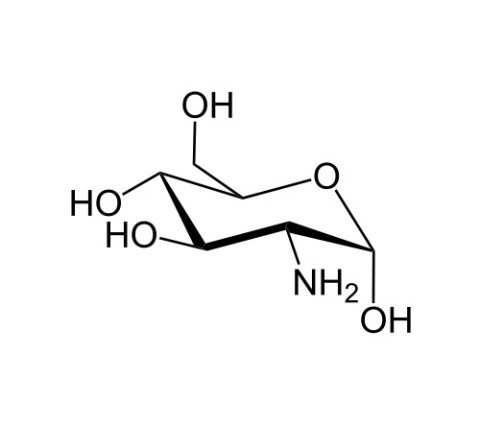
Glucosamine (C6H13NO5) is an amino sugar and a prominent precursor in the biochemical synthesis of glycosylated proteins and lipids. Glucosamine is part of the structure of the polysaccharides chitosan and chitin, which compose the exoskeletons of crustaceans and other arthropods, as well as the cell walls of fungi and many higher organisms. Glucosamine is one of the most abundant monosaccharides.[1] It is produced commercially by the hydrolysis of crustacean exoskeletons or, less commonly, by fermentation of a grain such as corn or wheat. Glucosamine appears to be safe for use as a dietary supplement; effectiveness has not been established for any condition. In the US it is one of the most common non-vitamin, non-mineral, dietary supplements used by adults.[2]
Starting at $28.00
Product Description
| IUPAC name
(3R,4R,5S)-3-Amino-6-(hydroxymethyl)oxane-2,4,5-triol
|
|
| Other names
2-Amino-2-deoxy-glucose
Chitosamine |
|
CAS Number
|
3416-24-8 |
|
Beilstein Reference
|
1723616 |
| ChEBI | CHEBI:5417 |
| ChEMBL | ChEMBL181132 |
| ChemSpider | 388352 |
| DrugBank | DB01296 |
| EC Number | 222-311-2 |
|
Gmelin Reference
|
720725 |
| Jmol interactive 3D | Image |
| KEGG | D04334 |
| MeSH | Glucosamine |
| PubChem | 439213 |
| UNII | N08U5BOQ1K |
|
InChI[show]
|
|
|
SMILES[show]
|
|
|
Chemical formula
|
C6H13NO5 |
| Molar mass | 179.17 g·mol−1 |
| Density | 1.563 g/mL |
| Melting point | 150 °C (302 °F; 423 K) |
| log P | -2.175 |
| Acidity (pKa) | 12.273 |
| Basicity (pKb) | 1.724 |
| Pharmacology | |
|---|---|
| ATC code | M01AX05 |