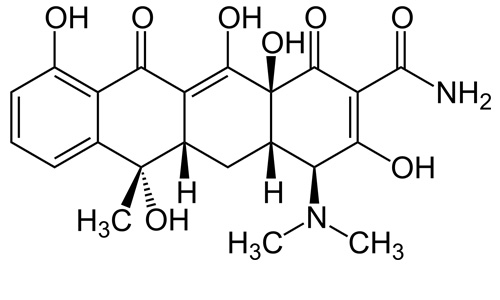
Tetracycline Hcl
Tetracycline is an antibiotic used to treat a number of bacterial infections. It is commonly used to treat acne and rosacea. Historically it was important in reducing the number of deaths from cholera.
It is broad-spectrum and of the polyketide class. It is produced by the Streptomyces genus of Actinobacteria. It is a protein synthesis inhibitor.
It is on the World Health Organization's List of Essential Medicines, a list of the most important medication needed in a basic health system.[1] Tetracycline is marketed under the brand names Sumycin, Tetracyn, Lymecycline, and Panmycin, among others. Actisite is a thread-like fiber formulation used in dental applications. It is also used to produce several semisynthetic derivatives, which together are known as the tetracycline antibiotics. The term “tetracycline” is also used to denote the four-ring system of this compound; “tetracyclines” are related substances that contain the same four-ring system.

Starting at
Product Description
| Pronunciation | /ˌtɛtrəˈsaɪkliːn/ |
|---|---|
| Trade names | Sumycin |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a682098 |
| Licence data | US FDA:link |
| Pregnancy category |
|
| Legal status |
|
| Routes of administration |
oral, topical (skin and eye), intramuscular, intravenous |
| Bioavailability | 75% |
|---|---|
| Metabolism | Not metabolised |
| Biological half-life | 8-11 hours, 57-108 hours (kidney impairment) |
| Excretion | Urine (>60%), feces |
| CAS Number | 60-54-8 64-75-5 (hydrochloride) |
|---|---|
| ATC code | A01AB13 D06AA04 J01AA07 S01AA09 S02AA08 S03AA02 QG01AA90 QG51AA02 QJ51AA07 |
| PubChem | CID: 643969 |
| DrugBank | DB00759 |
| ChemSpider | 10257122 |
| UNII | F8VB5M810T |
| KEGG | D00201 |
| ChEBI | CHEBI:27902 |
| ChEMBL | CHEMBL1440 |
| Formula | C22H24N2O8 |
|---|---|
| Molecular mass | 444.435 g/mol |
|
SMILES[show] |
|
|
InChI[show] |
|