Welcome visitor you can
login or register
0 items - $0.00
No products in the cart.
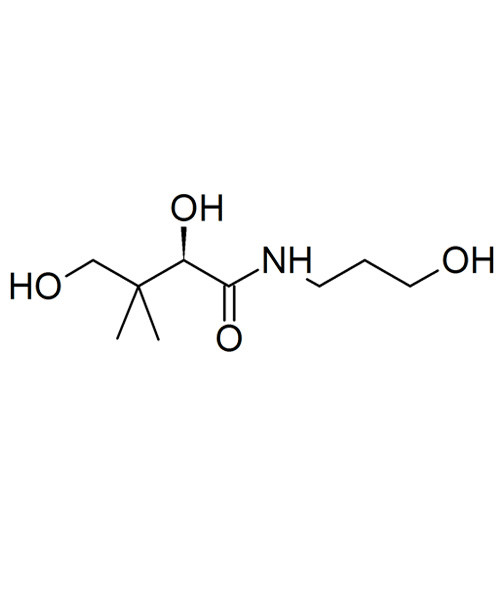
Vitamin B5 D Panthenol
Panthenol (pantothenol) is the alcohol analog of pantothenic acid (vitamin B5), and is thus a provitamin of B5. In organisms it is quickly oxidized to pantothenate [pantothenic acid]. Panthenol is a highly viscous transparent liquid at room temperature, but salts of pantothenic acid (for example sodium pantothenate) are powders (typically white). It is soluble in water, alcohol, propylene glycol, ether and chloroform, and slightly soluble in glycerin.
Panthenol comes in two enantiomers, D and L. Only D-panthenol (dexpanthenol) is biologically active, however both forms have moisturizing properties. For cosmetic use, panthenol comes either in D form, or as a racemic mixture of D and L (DL-panthenol).
SKU: n/a.
Category: Vitamins
Starting at $19.95
Product Description
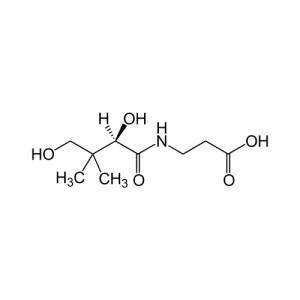
| IUPAC name 2,4-Dihydroxy-N-(3-hydroxypropyl)-3,3-dimethylbutanamide[1] |
|
| Other names Bepanthen Dexpanthenol |
| CAS Number | 16485-10-2 81-13-0 R |
| 3DMet | B00882 |
| Beilstein Reference | 1724945, 1724947 R |
| ChEBI | CHEBI:27373 |
| ChEMBL | ChEMBL1200979 |
| ChemSpider | 4516 115991 R 4677984 S |
| EC Number | 240-540-6 |
| Jmol interactive 3D | Image Image |
| KEGG | D03726 |
| MeSH | dexpanthenol |
| PubChem | 4678 131204 R 5748487 S |
| RTECS number | ES4316500 |
| UNII | 1O6C93RI7Z |
|
InChI[show] |
|
|
SMILES[show] |
|
| Chemical formula | C9H19NO4 |
| Molar mass | 205.25 g·mol−1 |
| Appearance | Colourless liquid[contradictory] |
| Density | 1.2 g mL−1 (at 20 °C) |
| Melting point | 66 to 69 °C (151 to 156 °F; 339 to 342 K) |
| log P | −0.989 |
| Acidity (pKa) | 13.033 |
| Basicity (pKb) | 0.964 |
| Refractive index (nD) | 1.499 |
| ATC code | A11HA30 D03AX03, S01XA12 |
| NFPA 704 |  |
| Lethal dose or concentration (LD, LC): | |
| LD50 (Median dose) | 10,100 mg kg−1 (intraperitoneal, mouse); 15,000 mg kg−1 (oral, mouse) |
| Related compounds |
Arginine Theanine Pantothenic acid Hopantenic acid |