0 Carrito - $0.00
No products in the cart.
METIL SULFONIL METANO (MSM)
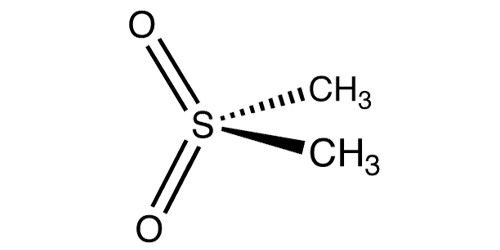
Methylsulfonylmethane (MSM) is an organosulfur compound with the formula (CH3)2SO2. It is also known by several other names including DMSO2, methyl sulfone, and dimethyl sulfone.[2] This colorless solid features the sulfonyl functional group and is considered relatively inert chemically. It occurs naturally in some primitive plants, is present in small amounts in many foods and beverages, and is marketed as a dietary supplement. It is also commonly found in the atmosphere above marine areas, where it is used as a carbon source by the airborne bacteria Afipia,[3] and is found distinctively in human melanoma cells.[4]
SKU: n/a.
Category: Nutricional
Starting at
Product added!
Add to wishlist
The product is already in the wishlist!
Add to wishlist
Product Description
| IUPAC name
dimethyl sulfone
|
|
| Preferred IUPAC name
methanesulfonylmethane
|
|
| Other names
methyl sulfone
methylsulfonylmethane sulfonylbismethane DMSO2 |
|
CAS Number
|
67-71-0 |
| ChEBI | CHEBI:9349 |
| ChEMBL | ChEMBL25028 |
| ChemSpider | 5978 |
| Jmol interactive 3D | Image |
| KEGG | C11142 |
| PubChem | 6213 |
| RTECS number | PB2785000 |
| UNII | 9H4PO4Z4FT |
|
InChI[show]
|
|
|
SMILES[show]
|
|
|
Chemical formula
|
C2H6O2S |
| Molar mass | 94.13 g·mol−1 |
| Appearance | White crystalline solid |
| Density | 1.45 g/cm3 |
| Melting point | 109 °C (228 °F; 382 K) |
| Boiling point | 248[1] °C (478 °F; 521 K) |
| Safety data sheet | External MSDS |
| S-phrases | S22 S24/25 |
| NFPA 704 |  |
| Flash point | 143 °C (289 °F; 416 K) |
|
Related compounds
|
DMSO dimethyl sulfide dimethyl sulfate sulfolane |
Be the first to review “METIL SULFONIL METANO (MSM)” Cancel reply
Related Products
Product added!
Add to wishlist
The product is already in the wishlist!
Add to wishlist
ACIDO ALFALIPOICO

Product added!
Add to wishlist
The product is already in the wishlist!
Add to wishlist
OMEGA 9

Product added!
Add to wishlist
The product is already in the wishlist!
Add to wishlist
OMEGA 3
Product added!
Add to wishlist
The product is already in the wishlist!
Add to wishlist

Reviews
There are no reviews yet.