Welcome visitor you can
login or register
0 items - $0.00
No products in the cart.
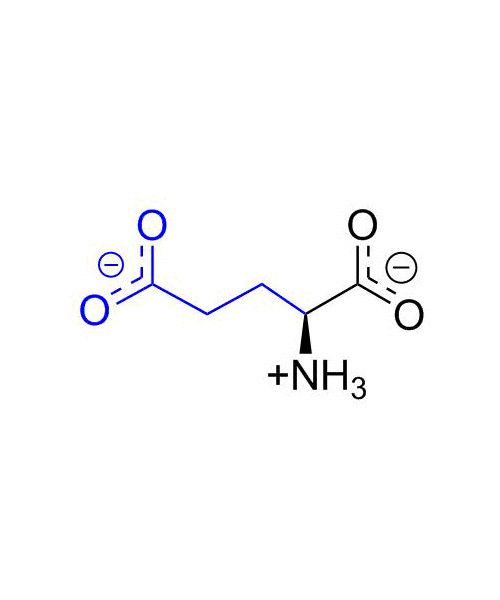
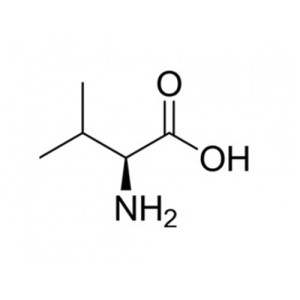
L- Glutamic Acid
Glutamic acid (abbreviated as Glu or E ; encoded by the codons GAA or GAG) is an ɑ-amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated -+NH3 form under biological conditions), an α-carboxylic acid group (which is in the deprotonated –COO- form under biological conditions), and a side chain carboxylic acid, classifying it as a polar negatively charged (at physiological pH), aliphatic amino acid. It is non-essential in humans, meaning the body can synthesize it.
SKU: n/a.
Category: Amino Acids
Starting at $5.53
Product Description
| Systematic IUPAC name
2-Aminopentanedioic acid
|
|
| Other names
2-Aminoglutaric acid
|
|
CAS Number
|
56-86-0 (L isomer) 617-65-2 (D/L racemate) |
| ChEBI | CHEBI:18237 |
| ChEMBL | ChEMBL276389. |
| ChemSpider | 591 |
| Jmol interactive 3D | Image |
| KEGG | D0434 |
| UNII | 61LJO5I15S |
|
InChI[show]
|
|
|
SMILES[show]
|
|
|
Chemical formula
|
C5H9NO4 |
| Molar mass | 147.13 g·mol−1 |
| Appearance | white crystalline powder |
| Density | 1.4601 (20 °C) |
| Melting point | 199 °C (390 °F; 472 K) decomposes |
|
Solubility in water
|
7.5 g/L (20 °C)[1] |
| Solubility | 0.00035g/100g ethanol (25 °C)[2] |
| Acidity (pKa) | 2.1, 4.07, 9.47 [3] |
| Safety data sheet | See: data page |
| NFPA 704 | 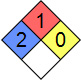 |
|
Structure and
properties |
Refractive index (n), Dielectric constant (εr), etc. |
|
Thermodynamic
data |
Phase behaviour solid–liquid–gas |
|
Spectral data
|
UV, IR, NMR, MS |