Welcome visitor you can
login or register
0 items - $0.00
No products in the cart.
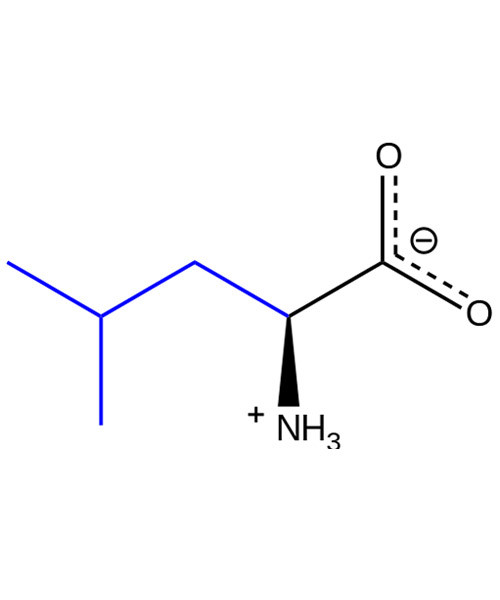
L-Leucine
Leucine (abbreviated as Leu or L; encoded by the six codons UUA, UUG, CUU, CUC, CUA, and CUG) is an ɑ-amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated -+NH3 form under biological conditions), an α-carboxylic acid group (which is in the deprotonated –COO- form under biological conditions), and an isobutyl side chain, classifying it as a nonpolar (at physiological pH) amino acid. It is essential in humans, meaning the body cannot synthesize it and thus it must be obtained from the diet.
SKU: n/a.
Category: Amino Acids
Starting at $18.76
Product Description
| IUPAC name
Leucine
|
|
| Other names
2-Amino-4-methylpentanoic acid
|
|
CAS Number
|
61-90-5 |
| ChEBI | CHEBI:57427 |
| ChEMBL | ChEMBL291962 |
| ChemSpider | 5880 |
| DrugBank | DB01746 |
|
IUPHAR/BPS
|
3312 |
| Jmol interactive 3D | Image |
| KEGG | D00030 |
| PubChem | 6106 |
| UNII | GMW67QNF9C |
|
InChI[show]
|
|
|
SMILES[show]
|
|
|
Chemical formula
|
C6H13NO2 |
| Molar mass | 131.18 g·mol−1 |
| Acidity (pKa) | 2.36 (carboxyl), 9.60 (amino)[1] |
|
Structure and
properties |
Refractive index (n), Dielectric constant (εr), etc. |
|
Thermodynamic
data |
Phase behaviour solid–liquid–gas |
|
Spectral data
|
UV, IR, NMR, MS |