Welcome visitor you can
login or register
0 items - $0.00
No products in the cart.
Sorbitol (Liquid)
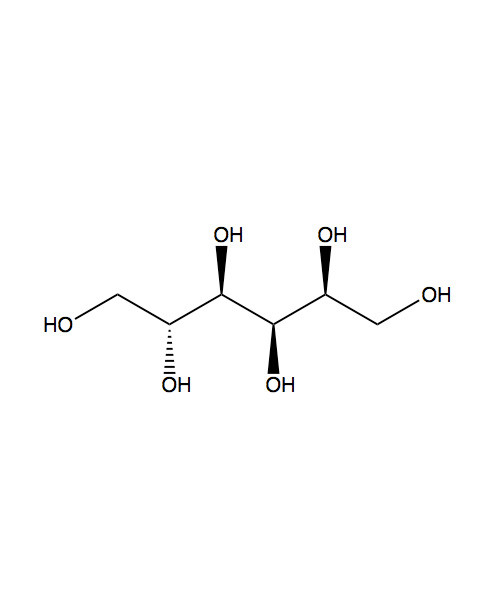
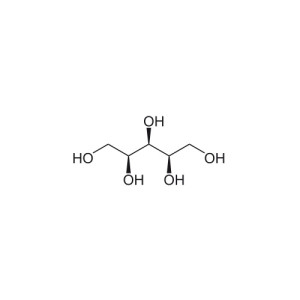
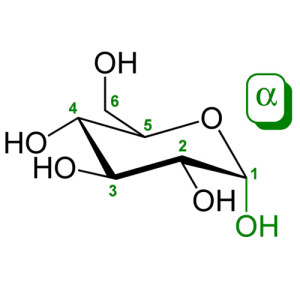
Sorbitol[pronunciation?], also known as glucitol[pronunciation?], is a sugar alcohol with a sweet taste which the human body metabolizes slowly. It can be obtained by reduction of glucose, changing the aldehyde group to a hydroxyl group. Most sorbitol is made from corn syrup, but it is also found in apples, pears, peaches, and prunes.[1] It is converted to fructose by sorbitol-6-phosphate 2-dehydrogenase. Sorbitol is an isomer of mannitol, another sugar alcohol; the two differ only in the orientation of the hydroxyl group on carbon 2.[2] While similar, the two sugar alcohols have very different sources in nature, melting points, and uses.

Make an enquiry for this product
Category: Sweetners
Starting at
Product Description
| IUPAC name (2S,3R,4R,5R)-Hexane-1,2,3,4,5,6-hexol |
|
| Other names D-glucitol; D-Sorbitol; Sorbogem; Sorbo |
| CAS Number | 50-70-4 |
| ChEBI | CHEBI:17924 |
| ChEMBL | ChEMBL1682 |
| ChemSpider | 5576 |
| DrugBank | DB01638 |
| Jmol interactive 3D | Image |
| MeSH | Sorbitol |
| PubChem | 5780 |
| UNII | 506T60A25R |
|
InChI[show] |
|
|
SMILES[show] |
|
| Chemical formula | C6H14O6 |
| Molar mass | 182.17 g/mol |
| Appearance | white crystalline powder |
| Density | 1.489 g/cm3 |
| Melting point | 111 °C (232 °F; 384 K) |
| Boiling point | 290–295 °C (554–563 °F; 563–568 K) |
| Solubility in water | 2350 g/L |
| ATC code | A06AD18 A06AG07 B05CX02 V04CC01 |
| NFPA 704 | 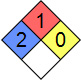 |
| Flash point | 100 °C (212 °F; 373 K) |
|
Autoignition
temperature |
150 °C (302 °F; 423 K) |